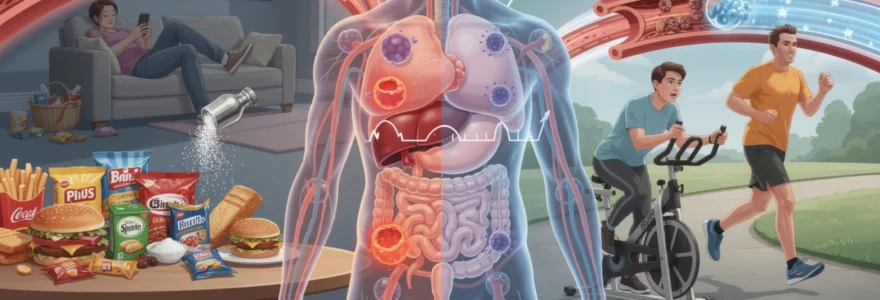
The decisions you make every day—what you eat, how much you move, whether you smoke, and how well you sleep—shape your long-term health in profound ways. Chronic diseases, which account for seven out of ten deaths globally and affect millions of people worldwide, are not simply inevitable consequences of aging or genetics. Research increasingly demonstrates that lifestyle factors play a dominant role in determining whether you develop conditions such as cardiovascular disease, type 2 diabetes, chronic obstructive pulmonary disease, and liver cirrhosis. Understanding the intricate biological mechanisms through which daily habits influence disease pathogenesis empowers you to make informed choices that can dramatically reduce your risk of chronic illness and enhance your overall wellbeing throughout your lifetime.
Dietary patterns and their direct impact on cardiovascular disease pathogenesis
The food you consume directly influences the development and progression of cardiovascular disease through multiple biological pathways. Your dietary choices affect everything from blood vessel integrity to inflammatory markers, cholesterol profiles, and blood pressure regulation. The relationship between nutrition and heart health extends far beyond simple calorie counting—it involves complex biochemical interactions that either protect or damage your cardiovascular system over time.
Excessive sodium intake and Hypertension-Induced arterial damage
When you consume excessive amounts of sodium—particularly from processed foods and restaurant meals—your body retains additional fluid to maintain proper electrolyte balance. This increased fluid volume forces your heart to work harder, elevating blood pressure and placing mechanical stress on arterial walls. Over time, this sustained pressure damages the delicate endothelial lining of blood vessels, creating microscopic tears where cholesterol and other substances can accumulate. The World Health Organization recommends limiting sodium intake to less than 2,000 milligrams daily, yet the average person consumes nearly double this amount, contributing to the global hypertension epidemic affecting over 1.28 billion adults worldwide.
Trans fats and Low-Density lipoprotein cholesterol accumulation
Trans fats, found predominantly in partially hydrogenated oils used in many baked goods and fried foods, represent one of the most harmful dietary components for cardiovascular health. These artificially modified fats simultaneously increase your low-density lipoprotein (LDL) cholesterol—often called “bad” cholesterol—while decreasing high-density lipoprotein (HDL) cholesterol, which helps remove harmful cholesterol from your bloodstream. This dual mechanism creates an especially dangerous lipid profile that accelerates atherosclerotic plaque formation in your arteries. Even small amounts of trans fats can significantly impact your cardiovascular risk, which is why many countries have implemented regulations to eliminate these substances from the food supply.
Mediterranean diet adherence and reduced atherosclerotic plaque formation
In contrast to harmful dietary patterns, the Mediterranean diet demonstrates remarkable protective effects against cardiovascular disease through multiple mechanisms. This eating pattern, characterized by abundant fruits, vegetables, whole grains, legumes, olive oil, and moderate fish consumption, provides high levels of antioxidants, fibre, and omega-3 fatty acids that reduce inflammation and oxidative stress in your blood vessels. Studies show that adherence to Mediterranean dietary principles can reduce cardiovascular disease risk by up to 30%, with benefits extending to stroke prevention, improved cholesterol profiles, and better blood sugar control. The anti-inflammatory compounds in olive oil, particularly oleocanthal, work similarly to low-dose aspirin in protecting your cardiovascular system from damage.
Ultra-processed food consumption and endothelial dysfunction
Ultra-processed foods—products containing ingredients you would never use in home cooking, such as high-fructose corn syrup, artificial emulsifiers, and synthetic flavourings—comprise an increasingly large proportion of modern diets in developed countries. These foods trigger endothelial dysfunction, impairing your blood vessels’ ability to dilate properly in response to increased blood flow demands. The combination of excessive refined carbohydrates, unhealthy fats, and artificial additives creates a pro-inflammatory environment in your body that accelerates arterial aging. Research indicates that each 10% increase in ultra-processed food consumption correlates with a 12% increase in cardiovascular disease risk, highlighting the importance of choosing whole, minimally processed foods whenever possible.
Physical inactivity as a primary risk factor for metabolic syndrome
Just as your diet shapes the health of your arteries, your daily movement patterns profoundly influence your metabolic health. Physical inactivity disrupts the finely tuned systems that regulate blood sugar, lipid metabolism, and body fat distribution, setting the stage for metabolic syndrome—a cluster of conditions including abdominal obesity, high blood pressure, elevated blood sugar, high triglycerides, and low HDL cholesterol. This combination dramatically increases your risk of type 2 diabetes, cardiovascular disease, and stroke. According to recent estimates, more than one in four adults worldwide does not meet recommended physical activity levels, making sedentary behaviour one of the most modifiable drivers of chronic disease.
Sedentary behaviour and insulin resistance mechanisms
When you spend prolonged periods sitting—at a desk, in a car, or in front of a screen—your skeletal muscles contract less frequently, and their demand for glucose drops. As a result, glucose transporters on muscle cells become less active, and insulin has a harder time moving sugar out of your bloodstream and into cells where it can be used for energy. Over time, this reduced responsiveness to insulin, known as insulin resistance, forces your pancreas to produce more insulin to achieve the same effect. Eventually, this compensatory mechanism becomes unsustainable, leading to chronically elevated blood sugar and a markedly increased risk of type 2 diabetes and other metabolic diseases.
Visceral adiposity accumulation through reduced energy expenditure
Low levels of daily movement also contribute to an imbalance between the calories you consume and the calories you burn, promoting fat storage—particularly around your abdomen. This visceral fat, which accumulates deep inside your abdominal cavity and surrounds vital organs, is metabolically active and releases pro-inflammatory cytokines and hormones that worsen insulin resistance and elevate blood pressure. Unlike subcutaneous fat just under the skin, visceral adiposity is strongly linked to metabolic syndrome and cardiovascular disease. Even modest increases in physical activity, such as adding a daily 20–30 minute walk, can help shift this energy balance, reduce central fat deposition, and lower your risk of chronic metabolic disorders.
Mitochondrial dysfunction and type 2 diabetes mellitus progression
At the cellular level, physical inactivity impairs the function and number of mitochondria—the tiny “power plants” within your muscle cells that generate energy. Reduced mitochondrial density and efficiency mean your muscles burn less glucose and fat during rest and activity, further aggravating insulin resistance and weight gain. Over time, this mitochondrial dysfunction contributes to the progression from prediabetes to overt type 2 diabetes, as cells become increasingly unable to process incoming nutrients effectively. Regular physical activity, by contrast, stimulates mitochondrial biogenesis, improving your cells’ capacity to handle glucose and lipids and slowing or even reversing early metabolic damage.
Exercise-induced AMPK activation and glucose homeostasis
One of the most powerful ways that movement protects against chronic disease involves the activation of AMP-activated protein kinase (AMPK), a key cellular energy sensor. During exercise, as your muscles use up stored energy, AMPK switches on and signals your cells to increase glucose uptake, enhance fat oxidation, and improve insulin sensitivity. Think of AMPK as your body’s “metabolic reset button”—each bout of physical activity helps restore glucose homeostasis and counteracts the effects of sedentary time. Integrating at least 150 minutes of moderate-intensity exercise per week, such as brisk walking or cycling, can significantly reduce your risk of metabolic syndrome and type 2 diabetes, even if you have a strong family history of these conditions.
Tobacco smoking and chronic obstructive pulmonary disease development
While diet and physical activity shape your cardiovascular and metabolic health, tobacco smoking is one of the most potent lifestyle factors driving chronic respiratory disease. Chronic obstructive pulmonary disease (COPD), which includes emphysema and chronic bronchitis, is primarily caused by long-term exposure to cigarette smoke and, increasingly, other nicotine products. COPD progressively limits airflow, making everyday activities like climbing stairs or walking short distances exhausting. Globally, smoking-related lung disease remains a leading cause of death and disability, yet it is largely preventable through smoking cessation and avoidance of secondhand smoke.
Nicotine-mediated inflammatory cytokine release in bronchial tissue
When you inhale cigarette smoke, thousands of chemicals, including nicotine, travel deep into your bronchial passages and lungs. Nicotine and other toxic compounds stimulate immune cells in your airway lining to release inflammatory cytokines, such as interleukin-6 and tumour necrosis factor-alpha. This chronic, low-grade inflammation thickens the bronchial walls, narrows the airways, and increases mucus production, all of which make breathing more difficult over time. Even in the early stages, this inflammatory response can reduce your lung function, and if smoking continues, it paves the way for irreversible structural damage characteristic of COPD.
Oxidative stress and alveolar destruction in emphysema
Cigarette smoke is also a potent source of free radicals—unstable molecules that cause oxidative stress and damage delicate lung tissue. The tiny air sacs in your lungs, called alveoli, are particularly vulnerable; they are responsible for oxygen exchange, and their thin walls can be likened to fragile soap bubbles. Prolonged exposure to smoke causes these walls to break down, leading to fewer, larger, and less efficient alveoli, a hallmark of emphysema. As this destruction progresses, your lungs lose their elasticity, trapping air and making it increasingly difficult to exhale fully, which is why people with advanced emphysema often feel short of breath even at rest.
Ciliary dysfunction and mucus hypersecretion in chronic bronchitis
In healthy lungs, tiny hair-like structures called cilia line the airways and work like conveyor belts to move mucus and trapped particles out of your respiratory tract. Smoking paralyzes and eventually damages these cilia, drastically reducing their ability to clear irritants and pathogens. At the same time, the goblet cells in your bronchial lining ramp up mucus production in response to chronic irritation, leading to thick, persistent phlegm. This combination of ciliary dysfunction and mucus hypersecretion defines chronic bronchitis and sets the stage for recurrent respiratory infections that further accelerate COPD progression.
Alcohol consumption patterns and hepatic cirrhosis progression
Alcohol intake is another critical lifestyle factor influencing the development of chronic liver disease. While occasional, moderate drinking may not cause significant harm in many individuals, regular heavy consumption or binge drinking patterns can initiate a cascade of liver damage that culminates in hepatic cirrhosis. In the early stages, excessive alcohol intake leads to fatty liver, where fat accumulates within liver cells and impairs their function. If the offending behaviour continues, inflammation (alcoholic hepatitis) and fibrosis follow, eventually resulting in widespread scarring that distorts liver architecture and compromises essential functions like detoxification, nutrient metabolism, and hormone regulation.
At the cellular level, the liver metabolizes alcohol primarily through the enzymes alcohol dehydrogenase and cytochrome P450 2E1, producing acetaldehyde, a highly toxic intermediate. Acetaldehyde directly damages liver cell membranes and proteins and promotes the formation of reactive oxygen species, driving oxidative stress. This ongoing injury activates hepatic stellate cells, which begin depositing collagen and other extracellular matrix components, thickening and stiffening liver tissue over time. Once cirrhosis develops, the damage is largely irreversible, and complications such as portal hypertension, liver failure, and hepatocellular carcinoma become much more likely.
However, your lifestyle choices can substantially alter this trajectory. Reducing alcohol intake, spacing drinks over time, and incorporating alcohol-free days each week give your liver an opportunity to recover from transient insults. For individuals already showing signs of fatty liver or early fibrosis, complete cessation of alcohol can halt or partially reverse damage, particularly when combined with a nutrient-dense diet and regular physical activity. If you find it difficult to cut back despite wanting to, seeking support from healthcare professionals, counselling, or structured programs can be a decisive step in protecting your long-term liver health.
Chronic sleep deprivation and neuroinflammatory disease pathways
Sleep is often overlooked when we think about lifestyle and chronic disease, yet it plays a foundational role in maintaining brain and body health. Chronic sleep deprivation—regularly getting fewer than seven hours of sleep per night for adults—disrupts hormonal balance, impairs cognitive function, and triggers inflammatory processes that affect multiple organ systems. Emerging research links poor sleep quality and short sleep duration to increased risks of neurodegenerative diseases, depression, obesity, cardiovascular disease, and impaired immune function. In many ways, adequate sleep functions as a nightly “reset” for your nervous and immune systems, helping to clear metabolic waste and regulate inflammatory signalling.
Within the brain, one of the key mechanisms involves the glymphatic system, a waste clearance network that becomes particularly active during deep sleep. This system helps remove neurotoxic proteins such as beta-amyloid and tau, which are implicated in Alzheimer’s disease and other forms of dementia. When you consistently cut your sleep short, this cleaning process becomes less efficient, allowing these proteins and other metabolites to accumulate and contribute to neuroinflammation. Chronic activation of microglia—the brain’s resident immune cells—further amplifies this inflammatory state, gradually damaging neurons and synapses and increasing the risk of cognitive decline over time.
Sleep deprivation also disrupts the delicate balance of hormones that regulate appetite, stress, and immune responses, including cortisol, leptin, and ghrelin. Elevated evening cortisol and altered circadian rhythms can increase systemic inflammation and exacerbate conditions such as hypertension, insulin resistance, and autoimmune disease. From a practical perspective, prioritizing consistent sleep routines—maintaining a regular bedtime, limiting screen exposure before bed, and creating a dark, quiet sleeping environment—can significantly improve sleep quality. If you struggle with chronic insomnia or loud snoring and frequent awakenings, consulting a healthcare professional for evaluation of sleep disorders such as sleep apnea is an important step in protecting both brain health and overall chronic disease risk.
Psychosocial stress and hypothalamic-pituitary-adrenal axis dysregulation in autoimmune disorders
Beyond physical habits like diet, movement, and sleep, your emotional environment and stress levels also profoundly affect chronic disease development. Persistent psychosocial stress—whether from work pressure, caregiving responsibilities, financial strain, or social isolation—can dysregulate your hypothalamic-pituitary-adrenal (HPA) axis, the central stress response system. Under acute stress, short bursts of cortisol and adrenaline help you cope with immediate challenges. However, when stress becomes chronic, this system loses its normal rhythmicity, leading to either persistently elevated or blunted cortisol levels that disturb immune regulation.
This HPA axis dysregulation plays an important role in the onset and exacerbation of autoimmune disorders, such as rheumatoid arthritis, systemic lupus erythematosus, and multiple sclerosis. Cortisol normally acts as a brake on inflammation, but when its production is erratic or receptors become less responsive, immune cells may become overactive and begin attacking the body’s own tissues. Chronic stress also promotes the release of pro-inflammatory cytokines and shifts immune cell populations, tipping the balance toward autoimmunity in genetically susceptible individuals. You might notice this connection in everyday life when flare-ups of autoimmune symptoms coincide with particularly stressful periods.
Managing psychosocial stress does not mean eliminating challenges altogether, which is neither realistic nor necessary. Instead, it involves building resilience and adopting coping strategies that restore HPA axis balance and calm overactive immune responses. Regular physical activity, mindfulness meditation, cognitive behavioural techniques, and strong social support networks are all evidence-based approaches for reducing perceived stress and improving immune function. By recognizing stress as a tangible biological risk factor—rather than just an emotional state—you can prioritize mental health practices as integral components of your strategy to prevent or manage chronic autoimmune and inflammatory diseases.

 Good health cannot be bought, but rather is an asset that you must create and then maintain on a daily basis.
Good health cannot be bought, but rather is an asset that you must create and then maintain on a daily basis.
