A rugby tackle fifteen years ago. A skiing accident that seemed minor at the time. That recurring sensation of your knee “giving way” when you descend stairs. What many dismiss as normal wear often conceals a progressive deterioration pathway—one where initial ligament damage silently rewrites your joint’s mechanical future. Research demonstrates that the connection between early instability and late-stage arthritis isn’t coincidental: it’s a causal biomechanical sequence. Understanding this cascade matters because intervention windows exist, but they close.
The hidden connection: when stability loss becomes degeneration
Knee deterioration occurs when ligament damage creates joint instability, leading to abnormal cartilage loading and chronic inflammation. This biomechanical cascade progressively destroys the shock-absorbing cartilage, eventually causing arthritis. Unlike simple wear from ageing, this process often follows a predictable pathway that begins with a specific injury event.
The conventional narrative separates sports injuries from arthritis. A torn anterior cruciate ligament belongs to athletic mishaps; osteoarthritis gets filed under inevitable ageing. Yet clinical evidence challenges this compartmentalisation. According to data from the World Health Organisation, joint injury—including fractures, strains, and repeated stress—is explicitly recognised as a known risk factor for developing osteoarthritis. The knee, with 365 million cases globally, represents the most frequently affected joint.
What you need to understand about knee deterioration
Four realities about knee deterioration you need to grasp before reading further:
- Ligament injuries create mechanical instability that directly accelerates cartilage breakdown through abnormal loading patterns
- Inflammation from initial trauma triggers enzymatic processes that continue destroying joint tissue long after the acute injury heals
- Post-traumatic osteoarthritis develops in approximately one quarter of patients within a decade following ligament tears
- Early specialist assessment combined with targeted rehabilitation can significantly alter the progression trajectory, but effectiveness diminishes with delay
What transforms a healed ligament tear into decades-long degeneration? The answer lies in residual mechanical disruption. When ligaments fail to restore their original tension and proprioceptive function, the joint loses its ability to distribute forces evenly across cartilage surfaces. This isn’t theoretical: peer-reviewed research tracking patients longitudinally reveals measurable progression rates linked directly to initial instability.

Consider the timeline disconnect that misleads many. An anterior cruciate ligament reconstruction at age 28 feels resolved by 30, yet inflammatory mediators persist in synovial fluid, cartilage experiences microtrauma from altered loading, and the joint shifts towards catabolism. By age 40, seemingly unrelated stiffness emerges—a direct descendant of that initial trauma. The distinction between reversible and irreversible damage matters profoundly: acute ligament tears can resolve with proper intervention, but chronic instability—abnormal movement repeated thousands of times monthly—etches itself into cartilage structure. Early recognition preserves tissue; delayed acknowledgement allows progressive erosion that no therapy can reverse.
The biomechanical cascade: from instability to cartilage breakdown
The progression from ligament injury to arthritis follows a three-stage biological sequence, each phase characterised by distinct pathological processes that compound into irreversible joint damage. Understanding these stages clarifies why timing interventions appropriately can alter outcomes.
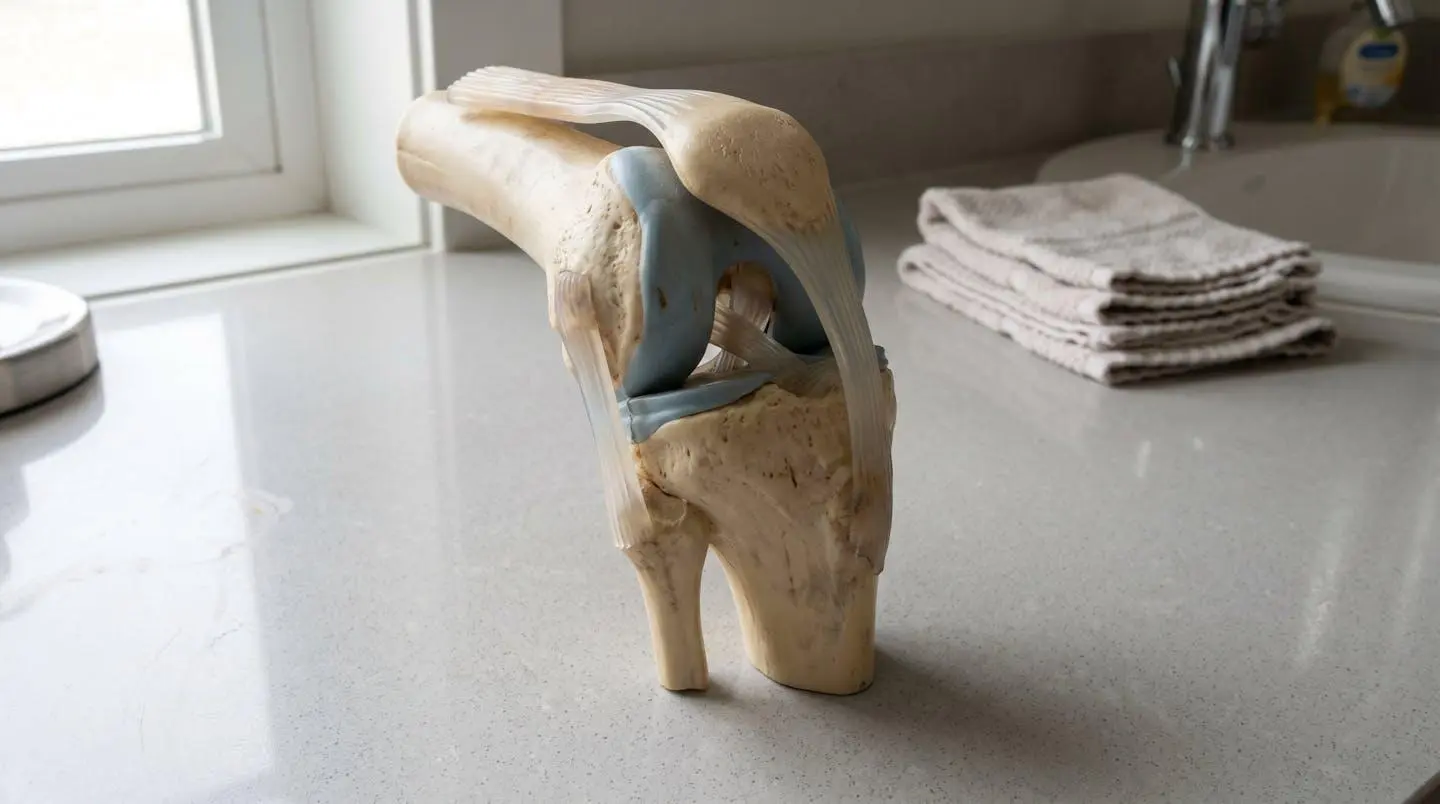
Ligament disruption immediately compromises the joint’s mechanical equilibrium. The anterior cruciate ligament, for instance, prevents excessive anterior translation of the tibia relative to the femur whilst controlling rotational stability. When torn, even partially, this restraint vanishes. The femur and tibia no longer move in their designed kinematic pathway. Instead, shear forces—forces that slide opposing surfaces against each other rather than compressing them—appear where they shouldn’t exist.
Cartilage evolved to withstand enormous compressive loads but possesses limited tolerance for shear stress. The collagen matrix within cartilage begins experiencing microtears at the molecular level. Surface fibrillation—the roughening of previously smooth cartilage—initiates within months of unaddressed instability, detectable on imaging before symptoms manifest.
The joint’s biological response to mechanical injury paradoxically accelerates damage. Chondrocytes—cartilage cells—detect abnormal stress through mechanoreceptors and release inflammatory cytokines including interleukin-1 and tumour necrosis factor-alpha. These molecules activate matrix metalloproteinases, enzymes that digest cartilage structure from within. What begins as a protective signalling cascade becomes a self-perpetuating destruction cycle.
Synovial inflammation compounds the problem. The joint lining, irritated by cartilage debris particles and inflammatory signals, thickens and releases additional degradative enzymes into synovial fluid. This fluid, normally a pristine lubricant, transforms into a corrosive soup bathing cartilage surfaces. Patients at this stage often report intermittent swelling—the knee that “comes and goes” in size—reflecting fluctuating inflammatory activity.
For those experiencing persistent knee symptoms following ligament injury, consulting a knee and orthopaedic surgeon enables comprehensive assessment of joint stability and inflammatory status through clinical examination combined with advanced imaging. This specialist evaluation distinguishes between reversible inflammatory processes and established structural damage, guiding whether conservative rehabilitation or surgical intervention offers optimal outcomes. Specialists can identify early-stage cartilage changes that remain treatable, whereas delayed evaluation may reveal progression beyond conservative management thresholds.
Cartilage possesses severely limited regenerative capacity due to its avascular nature—no blood supply means no delivery of repair cells or nutrients. As detailed in this peer-reviewed analysis published on PubMed Central, post-traumatic osteoarthritis develops in 9–12% of patients at five years and 23% at ten years following injuries causing joint instability such as anterior cruciate ligament tears. These aren’t abstract statistics: they represent measurable cartilage volume loss, subchondral bone remodelling, and osteophyte formation visible on imaging.
The deterioration becomes self-accelerating. Damaged cartilage generates more inflammatory debris, which drives more enzymatic degradation, which creates additional damage. Simultaneously, altered loading patterns cause subchondral bone—the layer beneath cartilage—to remodel abnormally, becoming sclerotic in high-stress zones and cystic in others. This bone change further disrupts force distribution, creating a vicious cycle where mechanical and biological factors reinforce each other’s destructive effects.
The timeline below maps this three-stage cascade with clinical intervention windows for each phase, clarifying when specific actions remain effective versus when tissue damage becomes irreversible.
-
Acute inflammatory phase: Initial tissue damage triggers inflammatory cascade with cytokine release; critical rehabilitation window when neuromuscular retraining remains most effective
-
Chronic instability phase: Abnormal loading patterns stress cartilage surfaces; neuromuscular compensation develops but cannot fully restore normal kinematics; surface fibrillation detectable on imaging
-
Degenerative arthritis onset: Cumulative cartilage loss becomes symptomatic; stiffness, crepitus, and functional limitations emerge; radiographic changes confirm osteoarthritis diagnosis
Critical distinction: Acute injuries heal through inflammatory resolution; chronic instability perpetuates inflammation indefinitely. The biological environment never returns to baseline, explaining why seemingly recovered knees harbour ongoing deterioration processes years after the initial trauma.
Critical factors accelerating knee deterioration
Deterioration rates vary dramatically between individuals. Epidemiological research identifies both modifiable and fixed risk factors that influence progression speed. Recognising which elements you can control focuses preventive efforts where they generate maximum impact.
The following comparison distinguishes factors amenable to intervention from those requiring acceptance and adaptation. This framework guides realistic expectations whilst highlighting genuine opportunities for slowing degeneration.
| Risk Factor | Modifiable? | Impact Level | Practical Action |
|---|---|---|---|
| Body weight excess | Yes | High | Each kilogramme reduction decreases knee loading by 3-4 kg during walking; weight management represents the single most evidence-backed mechanical intervention |
| Neuromuscular control deficits | Yes | High | Targeted physiotherapy programmes improve quadriceps timing and co-contraction patterns, compensating partially for ligament insufficiency |
| High-impact activity continuation | Yes | Medium | Substituting high-impact sports (running, jumping) with lower-impact alternatives (cycling, swimming) decreases cumulative cartilage stress whilst maintaining fitness |
| Meniscal tear presence | Partially | High | Combined ligament-meniscus injuries significantly worsen prognosis; meniscal preservation surgery when anatomically suitable reduces subsequent cartilage loss |
| Age at injury | No | Medium | Injuries sustained in teens or twenties create decades of cumulative exposure; emphasises importance of early stabilisation for younger patients |
| Genetic cartilage susceptibility | No | Variable | Family history of early arthritis suggests heightened vulnerability; whilst unchangeable, it informs more aggressive preventive approach |
As outlined in the clinical framework established by NICE for managing osteoarthritis, joint instability and abnormal biomechanical loading represent specific clinical scenarios warranting intervention. NICE guidance acknowledges instability as a distinct pathway requiring targeted management rather than generic arthritis protocols, validating the mechanistic understanding that instability-driven degeneration follows different rules from primary age-related osteoarthritis.

Critical warning on combined injuries: Simultaneous ligament and meniscal damage creates exponentially worse outcomes than isolated ligament tears. The meniscus distributes load across cartilage; its loss concentrates stress into smaller contact areas. Patients with combined injuries face significantly elevated arthritis rates—research suggests approximately double the isolated ACL tear risk within a decade—making early comprehensive surgical assessment essential rather than optional.
Evidence-based strategies to slow degeneration
Intervention effectiveness correlates inversely with delay. The biological processes detailed above suggest clear implication: stabilising mechanics and controlling inflammation early interrupts the cascade before irreversible cartilage loss accumulates. The following strategies align with deterioration stages.
Neuromuscular rehabilitation represents the cornerstone of early-stage management. Proprioceptive training—exercises that retrain the brain’s awareness of joint position—partially compensates for lost ligament mechanoreceptors. Quadriceps strengthening provides dynamic stabilisation, with the muscle literally substituting for absent ligamentous restraint during controlled movements.
Structured programmes typically span 12–16 weeks, progressing from basic weight-bearing exercises to sport-specific movement patterns. Compliance determines outcomes: intermittent effort yields intermittent protection. Research tracking rehabilitated patients demonstrates that those maintaining neuromuscular exercises long-term show measurably slower cartilage volume loss on serial MRI compared to those abandoning protocols after initial symptom improvement.
Clinical pattern: the cost of delayed intervention
A recreational footballer in their early thirties sustains an anterior cruciate ligament tear during a weekend match. Initial swelling settles within weeks. The knee feels “mostly fine” for daily activities. Recreational sport continues at reduced intensity. No specialist consultation occurs. Five years later, recurrent “giving way” episodes emerge during pivoting movements. By year seven, persistent stiffness appears. Imaging at year eight reveals established cartilage thinning with early osteophyte formation—changes representing irreversible tissue loss.
This pattern illustrates the silent progression period where subjective recovery masks ongoing mechanical damage. Early arthroscopic assessment would have identified residual instability quantifiable through stress testing. Surgical reconstruction or intensive neuromuscular rehabilitation initiated within the first year would have restored more normal kinematics, potentially preventing or significantly delaying the degenerative changes that became symptomatic only when cartilage damage reached critical thresholds.
Once surface cartilage changes appear on imaging but remain pre-arthritic, focus shifts towards controlling inflammatory flares and optimising mechanical environment. Load management doesn’t mean complete rest—cartilage requires intermittent compression for nutrient diffusion—but rather intelligent activity selection.
Swimming and cycling generate joint motion without impact loading. Resistance training builds muscle strength that protects cartilage, provided exercises avoid end-range knee flexion under heavy load. Interval training—alternating intensity rather than sustaining constant high stress—allows tissue recovery between loading cycles.
Pharmacological inflammation control remains controversial. Topical non-steroidal anti-inflammatory drugs demonstrate modest benefit for symptom management without systemic risks. The comprehensive approaches for fighting osteoarthritis extend beyond mechanical stabilisation to encompass nutritional strategies, inflammation management, and targeted exercise protocols tailored to individual deterioration stages.
When conservative measures fail to control symptoms or imaging demonstrates progressive cartilage loss despite optimal management, surgical consultation becomes appropriate. Modern orthopaedic practice offers a spectrum of joint-preserving procedures before considering replacement.
Arthroscopic techniques can address mechanical symptoms from torn meniscal fragments or loose cartilage. Osteotomy—surgical realignment of bone to shift load from damaged to preserved cartilage zones—suits younger patients with localised wear. Biological treatments including autologous chondrocyte implantation attempt cartilage regeneration in carefully selected cases, though long-term outcomes remain under investigation.
For readers without current symptoms seeking proactive protection, understanding the role of joint stability in exercise contexts prevents the initial ligament damage that triggers deterioration cascades. Proper movement mechanics, adequate warm-up protocols, and progressive training load increases reduce acute injury risk substantially.
- Acute injury with immediate instability or substantial swelling:
Urgent specialist assessment within 48–72 hours; significant structural damage likely requiring imaging and consideration of surgical stabilisation
- Recurrent episodes of knee “giving way” over several months:
Specialist consultation within 2–4 weeks; repetitive instability indicates inadequate dynamic control and probable ligamentous insufficiency requiring assessment for reconstruction
- Gradual-onset stiffness without instability sensations:
GP assessment initially; trial of physiotherapy for 6–8 weeks; specialist referral if symptoms persist or worsen despite conservative management
- Past injury exceeding five years with new symptoms emerging:
Specialist review to assess degenerative progression and evaluate joint preservation options whilst tissue remains salvageable; delayed assessment risks missing intervention windows
- Document any “giving way” episodes: frequency, triggering movements, and associated swelling—this pattern guides diagnostic assessment
- Initiate bodyweight optimisation if BMI exceeds 25—each kilogramme reduction measurably decreases joint loading
- Request GP referral to physiotherapy specialising in sports injuries for neuromuscular assessment—generic exercise advice proves insufficient for instability patterns
- Avoid delaying specialist orthopaedic consultation beyond three months if instability persists—intervention effectiveness diminishes as tissue changes progress
Your questions about knee health and degeneration
Can you reverse knee cartilage damage once deterioration has started?
Cartilage lacks blood supply, severely limiting regenerative capacity. Established cartilage loss cannot be reversed through any current treatment. However, progression can be slowed or halted through mechanical stabilisation and inflammation control. Early-stage surface changes may stabilise with optimal management, whereas advanced full-thickness defects progress despite intervention. This underscores why early action matters: you cannot recover lost tissue, but you can preserve remaining cartilage.
How do I distinguish serious knee instability from normal muscular weakness?
True ligamentous instability produces a sudden “giving way” sensation during weight-bearing, often accompanied by a feeling that bones are sliding abnormally within the joint. Muscular weakness causes gradual fatigue and reduced endurance but not sudden buckling. Instability typically worsens with pivoting movements or descending stairs. If you can reliably reproduce the sensation through specific movements, or if episodes occur unpredictably causing near-falls, ligamentous insufficiency is likely and warrants clinical assessment including stress testing.
Does running accelerate knee arthritis if I already have ligament damage?
Running on an unstable knee concentrates impact forces into abnormal contact patterns, accelerating cartilage wear. Evidence suggests that high-impact activity with compromised joint mechanics increases deterioration rates compared to lower-impact alternatives. However, complete immobility also harms cartilage through reduced nutrient diffusion. The solution involves activity modification: substituting running with cycling or swimming maintains fitness whilst reducing detrimental loading. If running remains important, ensuring adequate neuromuscular control through rehabilitation and limiting weekly mileage reduces cumulative damage.
What distinguishes ACL reconstruction from knee replacement surgery?
ACL reconstruction aims to restore joint stability by replacing the torn ligament, typically performed after acute injury in younger active individuals to prevent subsequent arthritis. Knee replacement addresses established arthritis by resurfacing damaged bone and cartilage with prosthetic components, generally reserved for older patients with severe degeneration causing substantial pain and functional limitation. These represent opposite ends of the intervention spectrum: reconstruction prevents deterioration, replacement manages deterioration’s end-stage consequences.
Can arthritis develop from ligament injury even if the initial trauma seemed minor?
Absolutely. Injury severity doesn’t correlate perfectly with deterioration risk. Partial ligament tears causing subtle instability can trigger the same biomechanical cascade as complete ruptures. The critical factor isn’t dramatic immediate symptoms but rather persistent mechanical abnormality. Many patients recall past knee injuries dismissed as “minor sprains” that never fully resolved, later developing arthritis in that precise joint. Symptom severity at injury tells you about acute tissue damage; long-term degeneration risk depends on whether normal biomechanics restore fully.
Knee deterioration following ligament injury represents a preventable pathway rather than inevitable fate. Timing matters profoundly: early stabilisation preserves tissue that later interventions cannot recover, whilst delayed intervention addresses consequences rather than causes.
- If you’ve sustained recent knee injury with ongoing instability: request urgent specialist referral rather than adopting “wait and see” approach that allows tissue damage to accumulate
- If past injury occurred years ago but new symptoms emerge: recognise these as potential deterioration signals warranting assessment, not dismissible age-related changes
- If currently asymptomatic but concerned about injury prevention: invest effort in neuromuscular training and movement quality—preventing initial ligament damage eliminates the entire deterioration cascade
Important considerations
- This article provides general information on knee degeneration mechanisms and does not replace personalised medical assessment
- Progression timelines and risk factors vary significantly between individuals based on injury type, age, and overall health
- Any persistent knee pain, instability, or functional limitation warrants professional orthopaedic evaluation
- For personalised clinical guidance, consult your GP (General Practitioner) or an orthopaedic surgeon specialising in knee conditions

 Good health cannot be bought, but rather is an asset that you must create and then maintain on a daily basis.
Good health cannot be bought, but rather is an asset that you must create and then maintain on a daily basis.
