Metabolic health represents one of the most significant determinants of longevity and quality of life, yet many people remain unaware of a critical factor that influences their metabolic function: skeletal muscle mass. Beyond its obvious role in movement and physical strength, muscle tissue operates as a sophisticated metabolic organ that profoundly impacts blood glucose regulation, lipid metabolism, and systemic inflammation. As research continues to illuminate the complex biochemical pathways through which muscle mass influences metabolic outcomes, it becomes increasingly clear that preserving and building lean tissue should be a cornerstone of preventive health strategies. The alarming rise in metabolic syndrome, type 2 diabetes, and cardiovascular disease across ageing populations correlates strongly with declining muscle mass—a relationship that demands urgent attention from both healthcare professionals and individuals seeking to optimise their healthspan.
Skeletal muscle as an endocrine organ: myokine secretion and systemic metabolic regulation
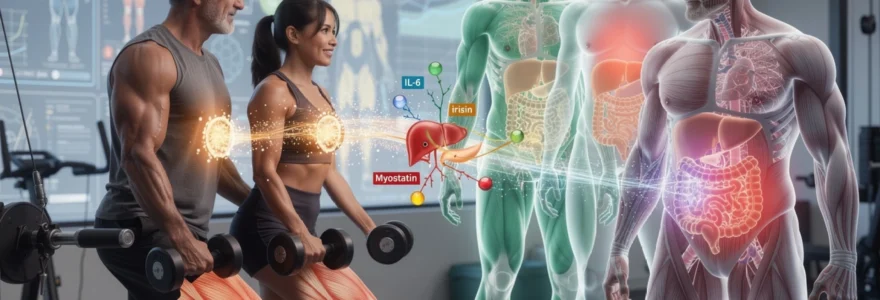
For decades, skeletal muscle was viewed primarily as a mechanical tissue responsible for locomotion and postural support. This perspective has fundamentally shifted as researchers have discovered that muscle functions as a dynamic endocrine organ, secreting dozens of bioactive proteins known as myokines. These signalling molecules facilitate cross-talk between muscle tissue and virtually every other organ system, including adipose tissue, liver, pancreas, brain, and cardiovascular system. When you engage in physical activity or maintain adequate muscle mass, your skeletal muscle releases these myokines into circulation, where they exert wide-ranging metabolic effects that extend far beyond local tissue adaptation.
The paradigm shift toward understanding muscle as an endocrine organ has profound implications for metabolic health management. Rather than viewing muscle mass simply as a reflection of physical fitness or aesthetic appearance, we must recognise it as a critical determinant of systemic metabolic function. Individuals with greater muscle mass possess enhanced capacity for myokine secretion, which translates to improved glucose homeostasis, reduced chronic inflammation, and better lipid profiles. This endocrine function of skeletal muscle represents a key mechanism through which resistance training and muscle preservation strategies confer metabolic benefits that persist even during periods of rest.
Interleukin-6 (IL-6) release during muscular contraction and glucose homeostasis
Among the various myokines secreted by contracting muscle fibres, interleukin-6 (IL-6) represents one of the most studied and metabolically significant. Unlike the chronic, low-grade IL-6 elevation associated with adipose tissue inflammation and metabolic dysfunction, the transient spike in IL-6 during and immediately following exercise exerts beneficial metabolic effects. During muscular contraction, IL-6 levels can increase up to 100-fold, promoting glucose uptake in working muscles through insulin-independent pathways and stimulating hepatic glucose output to maintain fuel supply during sustained physical activity.
The metabolic benefits of exercise-induced IL-6 extend beyond immediate glucose regulation. This myokine enhances fat oxidation, increases insulin sensitivity in peripheral tissues, and stimulates the secretion of anti-inflammatory cytokines that counteract metabolic dysfunction. Research demonstrates that individuals with greater muscle mass exhibit more robust IL-6 responses to exercise, suggesting that lean tissue quantity directly influences the magnitude of these beneficial metabolic adaptations. You can leverage this mechanism by engaging in regular resistance training, which not only stimulates acute IL-6 release but also increases the total muscle mass available to produce this critical myokine during future activity.
Irisin production from FNDC5 gene expression and adipose tissue browning
The discovery of irisin—a myokine derived from the cleavage of fibronectin type III domain-containing protein 5 (FNDC5)—has generated considerable excitement in metabolic research. Irisin levels increase in response to exercise, particularly activities that challenge muscle tissue and upregulate peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) expression. Once secreted into circulation, irisin travels to white adipose tissue, where it initiates a remarkable transformation process known as “browning,” converting metabolically inert white fat cells into metabolically active beige adipocytes that contain abundant mitochondria and express uncoupling protein 1 (UCP1).
This browning process increases whole-body energy expenditure, improves glucose handling, and reduces visceral fat accumulation: all key pillars of metabolic health. In both animal and human studies, higher irisin levels have been associated with better insulin sensitivity, lower body fat percentage, and improved lipid profiles. Crucially, maintaining or increasing muscle mass amplifies your capacity to produce irisin, meaning that regular resistance training not only builds lean tissue but also reshapes how your fat tissue behaves at rest. For individuals struggling with stubborn abdominal fat or early signs of insulin resistance, combining strength training with aerobic exercise offers a powerful way to harness irisin’s metabolic benefits and support long-term weight regulation.
Myostatin inhibition and its effects on insulin sensitivity pathways
Myostatin is often described as a “brake” on muscle growth: it is a myokine that inhibits muscle cell proliferation and differentiation, preventing excessive hypertrophy. While this regulatory role is essential in development, chronically elevated myostatin levels in adulthood are linked to reduced muscle mass, impaired strength, and unfavourable metabolic health. Elevated myostatin expression has been observed in people with obesity, type 2 diabetes, and non-alcoholic fatty liver disease, suggesting that excessive myostatin may contribute to both sarcopenia and metabolic dysfunction.
Reducing myostatin activity appears to improve several insulin sensitivity pathways. Experimental models in which myostatin is genetically deleted or pharmacologically blocked show increased skeletal muscle mass, reduced visceral adiposity, enhanced GLUT4 expression, and improved insulin-stimulated glucose uptake. In simpler terms, when the myostatin “brake” is eased off, muscles grow larger and more metabolically active, pulling more glucose out of the bloodstream with less insulin. Resistance training provides a physiological way to modulate myostatin: regular strength exercise downregulates myostatin expression in muscle while enhancing positive growth signals like insulin-like growth factor 1 (IGF-1). For you, this means that lifting weights does more than increase size and strength; it can help “re-tune” your muscle cells to respond better to insulin over time.
Brain-derived neurotrophic factor (BDNF) secretion and central metabolic control
Brain-derived neurotrophic factor (BDNF) is best known for its role in supporting neuronal growth, synaptic plasticity, and cognitive function. However, skeletal muscle also contributes to circulating BDNF, especially during and after exercise. This muscle-derived BDNF appears to act both locally, by enhancing fat oxidation in muscle fibres, and centrally, by influencing appetite regulation and energy balance via the hypothalamus. In this way, BDNF links muscular activity to central metabolic control, helping to coordinate how much energy you take in with how much you expend.
Higher BDNF levels are associated with improved glucose tolerance, reduced risk of metabolic syndrome, and better cardiovascular outcomes. Conversely, low BDNF has been observed in people with obesity, insulin resistance, and depressive symptoms—conditions that often coexist and reinforce each other. Building and preserving muscle mass through structured resistance training can support healthier BDNF dynamics, especially when combined with regular aerobic activity. For ageing adults concerned about both cognitive decline and metabolic disease, strength training offers a rare “two-for-one” intervention: it supports the brain while simultaneously enhancing peripheral metabolic health.
Sarcopenia-induced metabolic dysfunction: mechanisms of age-related muscle loss
Sarcopenia—the progressive loss of skeletal muscle mass and strength with age—is not just a mobility issue; it is a major driver of metabolic decline. From around the fourth decade of life, adults typically lose 1–2% of muscle mass and 1–3% of strength per year, with an accelerated decline after age 60. This loss of lean tissue reduces overall energy expenditure, impairs glucose disposal, and increases fat accumulation, particularly in visceral depots and within the liver. The end result is a higher risk of metabolic syndrome, type 2 diabetes, and cardiovascular events, even in individuals whose body weight has not changed dramatically.
Understanding why sarcopenia occurs is essential if we want to counteract its metabolic consequences. Age-related muscle loss is driven by a combination of mitochondrial dysfunction, impaired anabolic signalling (especially via the mTOR pathway), chronic low-grade inflammation, and reduced responsiveness to dietary protein. These mechanisms interact in a vicious cycle: as muscle quality declines, physical activity decreases, further worsening mitochondrial health and inflammatory status. The encouraging news is that targeted lifestyle strategies—chiefly resistance training, adequate protein intake, and inflammation control—can meaningfully slow, halt, or even partially reverse sarcopenia-induced metabolic dysfunction.
Mitochondrial dysfunction and reduced oxidative phosphorylation capacity in ageing muscle
Mitochondria, often described as the powerhouses of the cell, are particularly abundant in skeletal muscle, where they drive oxidative phosphorylation and ATP production. With ageing, mitochondrial number and function decline, leading to reduced capacity to oxidise glucose and fatty acids. This diminished oxidative capacity means that more energy substrates remain in circulation or get stored as fat, rather than being burned cleanly in muscle tissue. Over time, this contributes to elevated fasting glucose, higher triglyceride levels, and ectopic fat deposition within muscle and liver cells.
Clinically, ageing muscle with impaired mitochondrial function often appears weaker and more easily fatigued, and it may accumulate intramuscular fat—a phenomenon known as “myosteatosis.” This fat infiltration is not just cosmetic; it interferes with insulin signalling and further decreases muscle’s ability to act as an effective glucose sink. Fortunately, both endurance and resistance training can stimulate mitochondrial biogenesis and improve oxidative phosphorylation capacity, even in older adults. Think of exercise as a “software update” for your muscle cells: by challenging them regularly, you signal the body to repair and upgrade mitochondrial networks, thereby supporting better metabolic health well into later life.
Mammalian target of rapamycin (mTOR) pathway dysregulation and protein synthesis decline
The mammalian target of rapamycin (mTOR) pathway is a central regulator of muscle protein synthesis, responding to mechanical tension, amino acid availability (especially leucine), and hormonal signals such as insulin and IGF-1. In younger individuals, resistance exercise and adequate protein intake robustly activate mTOR, leading to efficient repair and growth of muscle fibres. With ageing, however, this signalling becomes blunted—a phenomenon sometimes called “anabolic resistance.” As a result, the same stimulus (a workout or a protein-rich meal) produces a smaller increase in muscle protein synthesis, tipping the balance toward net muscle loss over time.
Dysregulation of the mTOR pathway in ageing muscle contributes not only to structural decline but also to metabolic impairment. Reduced muscle protein turnover is associated with poorer glucose uptake, diminished glycogen storage, and increased fat infiltration in muscle tissue. Strategically, older adults can help re-sensitise the mTOR pathway through a combination of higher-quality resistance training (using progressive overload and near-failure sets) and optimised protein intake distributed across meals. For some individuals, especially those over 60, ensuring that each meal contains a sufficient dose of leucine-rich protein can make the difference between ongoing muscle loss and meaningful maintenance or growth.
Chronic low-grade inflammation and tumour necrosis factor-alpha (TNF-α) elevation
Ageing is often accompanied by a state of chronic low-grade inflammation, sometimes referred to as “inflammageing.” Elevated circulating levels of inflammatory cytokines such as tumour necrosis factor-alpha (TNF-α) and interleukin-1β (IL-1β) are common in older adults and individuals with obesity or metabolic syndrome. These cytokines directly impair muscle protein synthesis, promote proteolysis (muscle breakdown), and interfere with insulin signalling in skeletal muscle cells. Over time, this inflammatory milieu accelerates sarcopenia and contributes to worsening insulin resistance and dyslipidaemia.
TNF-α, in particular, has been shown to inhibit the insulin receptor substrate (IRS) pathway and reduce GLUT4 translocation to the muscle cell membrane, undermining the muscle’s ability to take up glucose after meals. From a practical perspective, this means that managing inflammation is a critical part of preserving both muscle mass and metabolic health as you age. Regular physical activity, sufficient sleep, weight management, and a dietary pattern rich in whole plant foods and omega-3 fats can all help reduce chronic inflammation. Resistance training plays a unique dual role here: it builds muscle mass and strength, while also shifting the inflammatory balance toward a more anti-inflammatory, myokine-rich profile.
Anabolic resistance to dietary protein and leucine supplementation strategies
One of the key challenges in combating sarcopenia is anabolic resistance: the reduced ability of ageing muscle to increase protein synthesis in response to dietary amino acids. Studies suggest that older adults may require a higher relative protein intake per meal to trigger the same anabolic response seen in younger individuals. Whereas 20 g of high-quality protein might be sufficient to stimulate maximal muscle protein synthesis in a young adult, someone over 65 may require 30–40 g, depending on body size and health status. Without this adjustment, even a seemingly “adequate” total daily protein intake may fail to preserve muscle mass.
Leucine, a branched-chain amino acid, plays a particularly important role in overcoming anabolic resistance because it acts as a powerful trigger for mTOR activation. Ensuring that each meal contains a leucine-rich protein source—such as whey, eggs, fish, poultry, or soy—can help older adults maximise the anabolic impact of their diet. In practice, you might aim for three to four evenly spaced meals per day, each delivering roughly 0.4–0.6 g of protein per kilogram of body weight, rather than concentrating most of your protein at dinner. In some cases, targeted leucine or essential amino acid supplements can be useful adjuncts, especially for those with reduced appetite or difficulty chewing high-protein foods.
GLUT4 translocation and insulin-independent glucose uptake in contracting muscle fibres
One of the most powerful ways skeletal muscle supports metabolic health is by clearing glucose from the bloodstream through GLUT4 transporters. At rest, a significant proportion of GLUT4 resides inside muscle cells, but in response to insulin or muscle contraction, these transporters move (or “translocate”) to the cell membrane, where they act as gateways for glucose uptake. During exercise, this process occurs largely through insulin-independent pathways, involving signalling molecules such as AMP-activated protein kinase (AMPK) and AS160, allowing muscles to pull in large amounts of glucose even when insulin levels are low or when tissues are insulin resistant.
Regular physical activity and higher muscle mass both increase the total pool of GLUT4 transporters available, enhancing your capacity to dispose of glucose after meals and stabilise blood sugar levels. This is one reason why even a single bout of exercise can improve insulin sensitivity for up to 24–48 hours, and why individuals with more lean mass tend to have lower fasting glucose and insulin levels. For people at risk of type 2 diabetes, incorporating resistance training and moderate-intensity aerobic sessions several times per week creates a “double hit”: it increases GLUT4 density and repeatedly activates contraction-mediated glucose uptake without relying solely on insulin. Over time, this can help reverse early insulin resistance and reduce the need for pharmacological intervention.
Visceral adiposity versus lean mass ratio: impact on metabolic syndrome biomarkers
When assessing metabolic health, focusing solely on body weight or body mass index (BMI) can be misleading. Two individuals with the same BMI can have vastly different risk profiles depending on their ratio of visceral adiposity (fat stored around internal organs) to lean mass. Visceral fat is strongly linked to elevated triglycerides, low HDL cholesterol, increased blood pressure, and systemic inflammation—all key components of metabolic syndrome. In contrast, higher lean mass acts as a metabolic buffer, improving glucose disposal, supporting favourable lipid profiles, and lowering inflammatory markers.
Clinical studies consistently show that people with higher muscle mass and lower visceral fat have better metabolic syndrome biomarkers, even if their total body weight is in the “overweight” range. This has given rise to the concept of “metabolically healthy obesity” and, conversely, “normal-weight obesity,” where individuals of normal BMI carry excess visceral fat and low lean mass, placing them at high cardiometabolic risk. For practitioners and patients alike, this underscores the importance of looking beyond the scale and prioritising strategies that reduce central fat while preserving or increasing skeletal muscle mass.
Waist-to-hip ratio and HOMA-IR index correlations in clinical studies
Simple anthropometric measurements can provide valuable insight into the balance between visceral adiposity and lean mass. Waist circumference and waist-to-hip ratio (WHR) are strongly associated with visceral fat stores and, in turn, with insulin resistance. The Homeostatic Model Assessment of Insulin Resistance (HOMA-IR), which uses fasting glucose and insulin levels, is often used in research and clinical practice to quantify insulin sensitivity. Numerous studies have demonstrated that higher WHR correlates with elevated HOMA-IR scores, particularly in individuals with low relative muscle mass.
Interestingly, the relationship between WHR and HOMA-IR is modified by lean mass: older adults or middle-aged individuals with higher muscle mass often show lower HOMA-IR at a given waist-to-hip ratio compared with those who are sarcopenic. This suggests that building and maintaining muscle can partially offset the metabolic burden of central adiposity, although it does not entirely neutralise the risks of excess visceral fat. Practically, you can track your waist circumference and, where possible, fasting glucose and insulin values to monitor trends in metabolic health over time. Combining these simple measures with resistance training and nutrition strategies targeting muscle growth provides a data-driven approach to improving insulin sensitivity and reducing the likelihood of progression to type 2 diabetes.
C-reactive protein (CRP) and adiponectin level variations with muscle mass changes
Beyond glucose and insulin, inflammatory and adipokine markers offer additional windows into the interplay between muscle mass, fat distribution, and metabolic health. High-sensitivity C-reactive protein (hs-CRP) is a widely used marker of systemic inflammation and is strongly predictive of cardiovascular risk. Adiponectin, on the other hand, is an anti-inflammatory adipokine that enhances insulin sensitivity and promotes fatty acid oxidation. Individuals with high visceral adiposity and low lean mass often present with elevated CRP and reduced adiponectin, a profile associated with metabolic syndrome and atherosclerosis.
Intervention studies show that increasing muscle mass through resistance training, especially when combined with modest fat loss, can lower CRP levels and raise adiponectin concentrations. Even in the absence of dramatic weight loss, improvements in body composition—shifting from fat mass to lean mass—are often reflected in more favourable CRP and adiponectin values. For you, this means that a well-structured strength-training programme can help “rebalance” inflammatory and adipokine signalling, reinforcing the improvements you see in blood glucose and lipid panels. If your healthcare provider tracks hs-CRP and adiponectin, watching these markers change over time can be a motivating way to see how your muscles are influencing your metabolic health from the inside out.
Dual-energy x-ray absorptiometry (DEXA) measurements for body composition assessment
To move beyond crude measures like BMI, clinicians and researchers increasingly rely on body composition tools such as dual-energy X-ray absorptiometry (DEXA). A DEXA scan provides a detailed breakdown of fat mass, lean mass, and bone mineral content, and can even distinguish between android (central) and gynoid (hip and thigh) fat distribution. This level of precision is especially valuable when monitoring the effects of interventions targeting metabolic health, because it reveals whether weight changes are coming from fat, muscle, or both.
For example, during a weight loss programme, a DEXA scan can highlight whether a person is losing a disproportionate amount of lean mass, which would predict a drop in resting metabolic rate and increased difficulty maintaining the weight loss. Conversely, improvements in total lean mass or regional muscle (such as in the legs and trunk) are often accompanied by better insulin sensitivity and cardiometabolic markers, even if total body weight remains stable. If you have access to DEXA testing through a medical clinic or performance centre, periodic scans—perhaps once or twice a year—can provide objective feedback on how effectively your training and nutrition strategies are shifting your body composition toward a more metabolically favourable profile.
Resistance training protocols and hypertrophic adaptations for metabolic health improvement
Not all resistance training programmes are created equal when it comes to building muscle mass and enhancing metabolic health. The most effective protocols for hypertrophy—and, by extension, for improving insulin sensitivity and energy expenditure—tend to share a few key characteristics: they involve multiple large muscle groups, use loads that feel challenging by the last few repetitions, and apply progressive overload over time. You do not need to train like a bodybuilder to reap these benefits, but you do need to give your muscles a consistent reason to adapt.
For most adults, especially those over 40 or with busy schedules, aiming for two to three full-body resistance sessions per week is a practical starting point. Each session might include compound movements such as squats or leg presses, hip hinges (like deadlifts or hip thrusts), pushing exercises (push-ups or bench presses), pulling movements (rows or pull-downs), and some core work. By performing 2–4 sets of 6–15 repetitions per exercise, and choosing loads that bring you within one to three reps of muscular fatigue, you stimulate both fast- and slow-twitch fibres and maximise the hypertrophic signal.
From a metabolic perspective, the goal is not just to gain muscle size, but to create muscle that is functionally active and insulin sensitive. This is where progressive overload and adequate recovery come in: over weeks and months, you gradually increase either the weight lifted, the number of repetitions, or the total number of sets, while ensuring sufficient rest between sessions to allow repair and growth. Combining this with a protein intake of roughly 1.2–1.6 g per kilogram of body weight per day will support muscle protein synthesis and minimise losses during periods of caloric restriction. If you are new to strength training or have joint concerns, working with a qualified coach or physiotherapist can help you tailor exercise selection and loading schemes to your specific needs, ensuring that you build metabolic-boosting muscle mass safely and sustainably.
Type II diabetes prevention through muscle mass preservation: evidence from framingham and UK biobank studies
Large-scale epidemiological studies provide compelling evidence that muscle mass preservation is a powerful tool for preventing type 2 diabetes. Data from cohorts such as the Framingham Heart Study and the UK Biobank have shown that individuals with higher relative lean mass—and particularly greater leg muscle mass—have significantly lower incidence of new-onset type 2 diabetes over follow-up periods of 5–15 years. These associations persist even after adjusting for traditional risk factors like age, BMI, smoking status, and baseline physical activity, underscoring the independent protective effect of skeletal muscle.
In some analyses, moving from the lowest to the highest quartile of muscle mass has been associated with reductions in diabetes risk of 30–60%, a magnitude comparable to that achieved with intensive lifestyle interventions focused on weight loss. Conversely, sarcopenia and rapid declines in muscle strength have been linked to higher HOMA-IR values, elevated fasting glucose, and more frequent progression from prediabetes to overt diabetes. These findings suggest that routine assessment of muscle mass and function—through grip strength, gait speed, or imaging-based measures—could enhance current diabetes risk prediction models and help clinicians identify patients who would benefit most from targeted resistance training and nutrition support.
For you, the takeaway is clear: protecting your muscle mass is not just about mobility or aesthetics; it is a potent, evidence-based strategy to reduce your lifetime risk of type 2 diabetes and related complications. By combining regular resistance training with adequate protein intake, attention to sleep and stress, and monitoring of key biomarkers such as waist circumference, fasting glucose, and hs-CRP, you can build a muscular “metabolic shield” that supports healthier ageing. As the research from Framingham, UK Biobank, and other cohorts continues to accumulate, one message stands out: muscle is medicine, and investing in it today pays substantial dividends for your metabolic health tomorrow.

 Good health cannot be bought, but rather is an asset that you must create and then maintain on a daily basis.
Good health cannot be bought, but rather is an asset that you must create and then maintain on a daily basis.
